Novo Nordisk recalls faulty cartridge holders in insulin pens due to potential health risks - YouTube

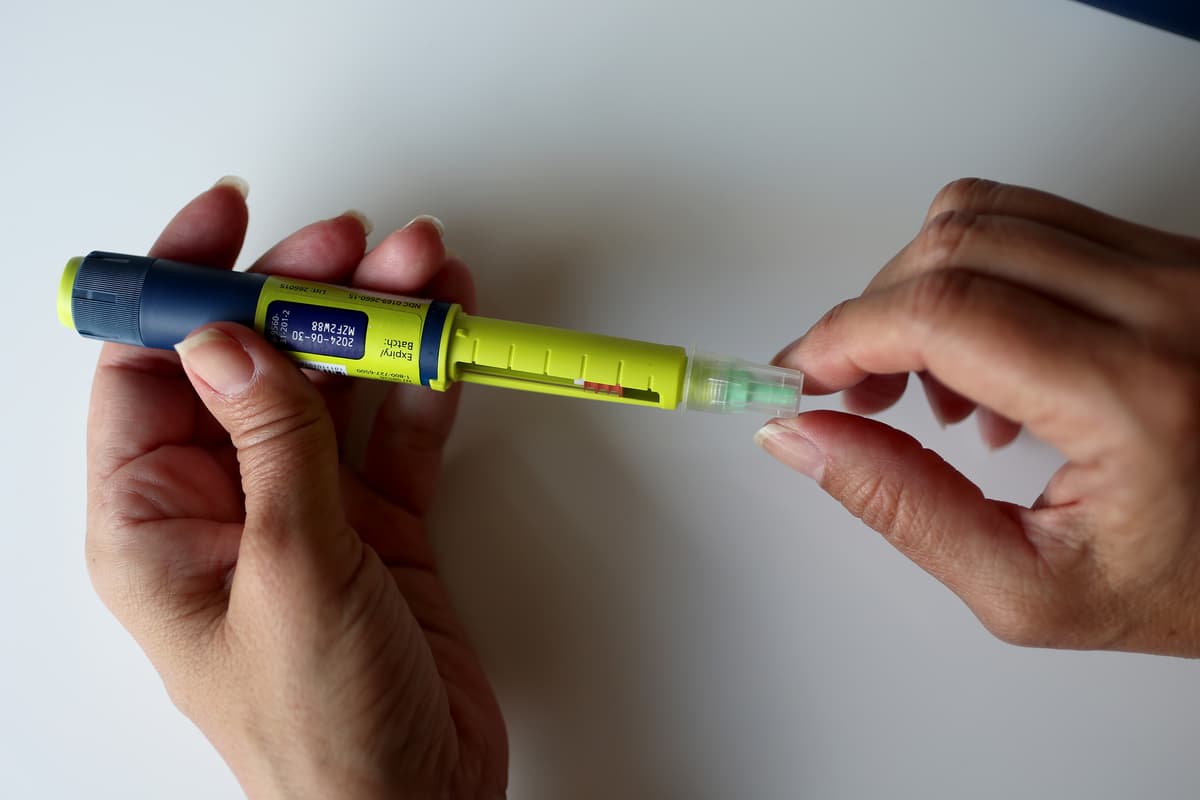
Recall: Novo Nordisk's Insulin Delivery Device Cartridge Holders Crack From Cleaning Agents - Pharmaceutical Processing World

Today, the FDA identified a Class I Recall, the most serious type of recall: Medtronic expanded its 2018 recall of remote controllers used with either... | By U.S. Food and Drug Administration | Facebook

Mylan Pharmaceuticals Inc., a Viatris Company, Issues Voluntary Nationwide Recall of One Batch of Insulin Glargine (Insulin glargine-yfgn) Injection Pens, 100 units/mL (U-100), Due to the Potential of Missing Labels on Some
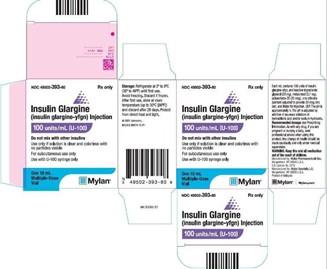
Mylan Pharmaceuticals Inc., a Viatris Company, Conducting Voluntary Nationwide Recall of One Batch of Insulin Glargine (Insulin glargine-yfgn) Injection, 100 units/mL (U-100), Due to the Potential for a Missing Label in the










/cloudfront-us-east-1.images.arcpublishing.com/gray/I352BZTZ2VIURCSSDSXGOCKRFY.jpg)