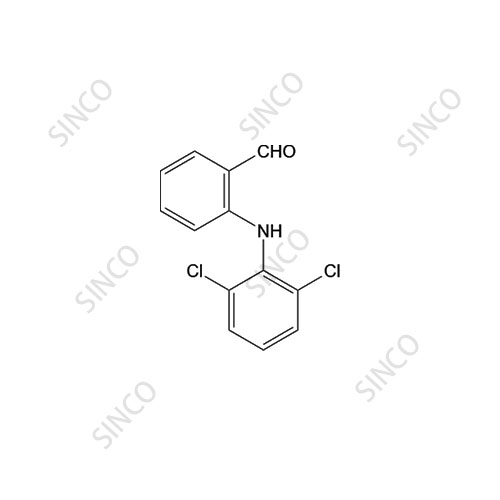
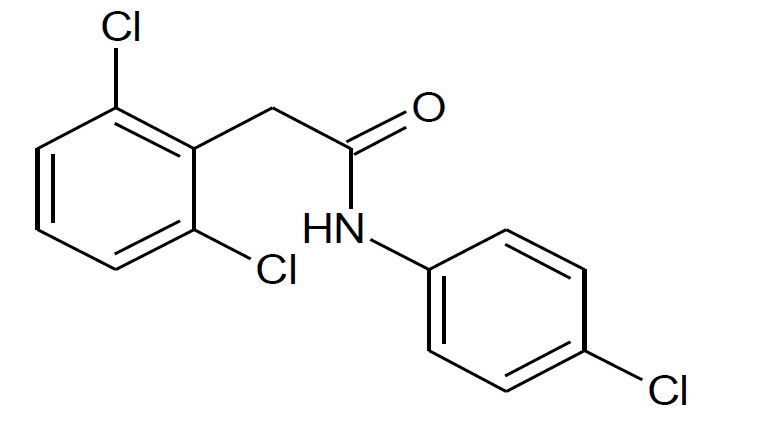
Diclofenac Impurity F Pharmaceutical Secondary Standard; Certified Reference Material | Sigma-Aldrich

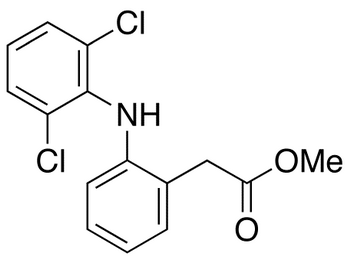
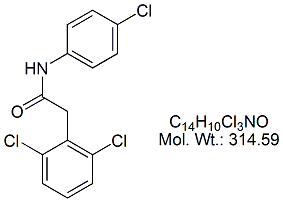
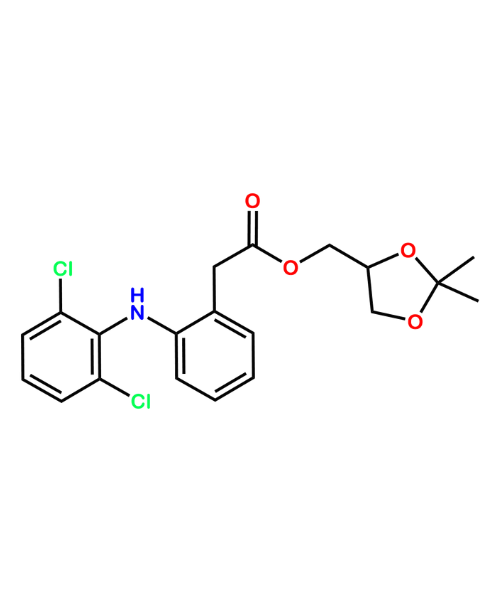
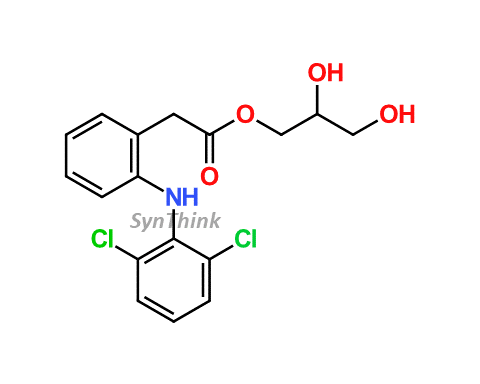
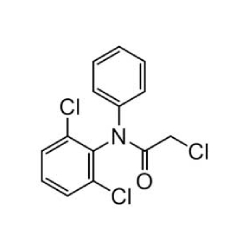
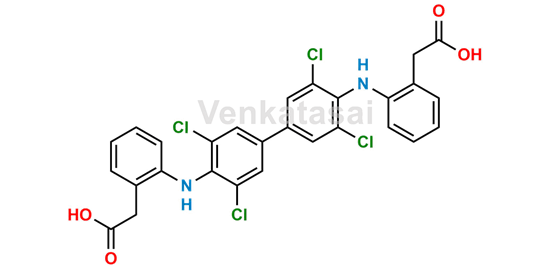
NAME OF THE PRODUCT : Diclofenac EP Impurity - F | NAME OF THE API IMPURITY STANDARDS : 2,6-Dichloro-N-(4-chlorophenyl)-benzeneacetamide | CAS Reg Number : 560075-65-2

Diclofenac Impurity F Pharmaceutical Secondary Standard; Certified Reference Material | Sigma-Aldrich